A few years back now, I received a very appropriate gift from The Girlfriend’s Sprog, in the form of a ‘Bio Orb‘* – a self-sustaining aquarium containing bioluminescent dinoflagellates. These are nearly microscopic phytoplankton that, at night, glow briefly brilliant blue when disturbed, and I say this was appropriate because I’ve had personal experience with them, back when I lived in Florida; during high summer, the Indian River Lagoon would play host to them, providing a fantastic natural light show at times.
A few years back now, I received a very appropriate gift from The Girlfriend’s Sprog, in the form of a ‘Bio Orb‘* – a self-sustaining aquarium containing bioluminescent dinoflagellates. These are nearly microscopic phytoplankton that, at night, glow briefly brilliant blue when disturbed, and I say this was appropriate because I’ve had personal experience with them, back when I lived in Florida; during high summer, the Indian River Lagoon would play host to them, providing a fantastic natural light show at times.
The great thing about the orb is that it’s, as I said, self-sustaining. I mean really. Dinoflagellates need very little to survive, and the globe only needs to be placed in moderate temperatures with some, but not too much, sunlight. Food is provided in the kit, but I’ve only fed them twice, because it’s only needed if there seems to be a die-off. This happened just once, as the migration of the sun through the year caused it to shine more directly into the window where I kept the orb, only in the morning, but it was a little too much. I shifted the orb back a little and everything has been going on its own since then, and that was probably two years ago now. From time to time, I pick up the globe at night and give it a little swish to see the burst of dim light that ensues. It’s pretty damn cool.
I did some initial experiments back then, mostly under the microscope, and managed to get a few photos of the individual organisms themselves. There’s really not a lot to them.
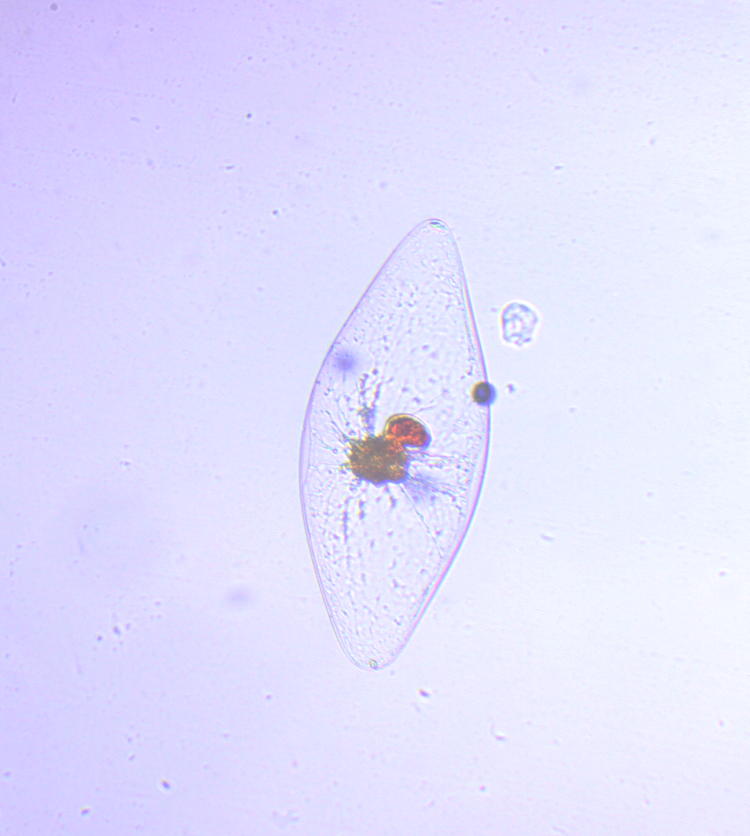
To be specific, this is a Pyrocystis fusiformis, a marine algae, one of the thousands of organisms known as phytoplankton. Length is less than a millimeter; I have a slide with a measuring scale etched into it, but finding one of these critters in the water sample can be tricky enough – somehow maneuvering it alongside the scale is a headache I have not yet tried to induce upon myself. This particular one, by the way, might be in the process of reproduction, since that nucleus appears to be splitting.
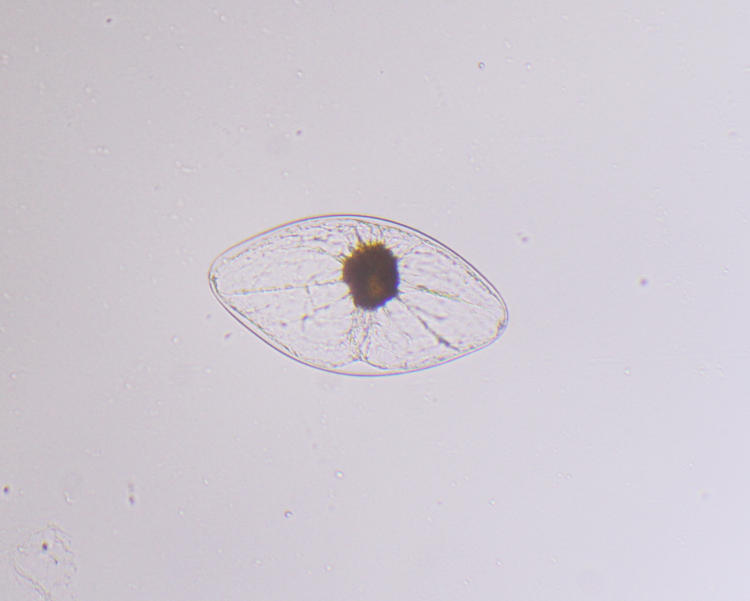
I also have to add another shot obtained at that time, which I pondered over.

When first trying to see the dinoflagellates, I found this and did a couple of shots with polarized light (thus the colors,) before I realized this was lint from the tissue that I used to clean the slide…
Anyway, as intimated in the previous post, I decided to tackle this again, getting the microscope back out, but also prepping some new equipment.
Microscopes, by nature, have an extremely short depth-of-field, so much so that they will often only get a portion of the subject in focus at once (which you’re going to see shortly.) One of these days I’ll start playing around seriously with image stacking, taking several photos and combining the sharpest portions of each into a new image. There’s software for this, I just haven’t messed with it yet, except manually.

I have a simple lensless adapter for the camera to fit onto the eyepiece of the microscope, but the eyepiece lens is removed. Due to this, the camera doesn’t get the same magnification (full-frame, anyway) that I can see, and focus always has to be readjusted. This image is with the 40X objective lens, but is less than 40X – how much so, maybe I’ll calculate someday.
But what I’d really like to capture is the actual bioluminescence. To say that this is tricky is putting it mildly, since it’s dim, within the long-exposure category but of course it doesn’t last anywhere near that long. Once while in Florida, I picked a good night when the dinos were active and set the camera up in the shallows of the lagoon, aiming down into the water (the camera remained dry, relax.) I then locked the shutter open and swished my hand around madly in front of the lens for a solid minute, just to see if I could get something to register. First off, let me tell you that swishing your hand madly in the water for 60 seconds straight is quite tiring. I’ve looked at the negatives a couple of times since then, and I managed enough exposure to delineate the edges of the frame, but nothing more than that.
The 7D, however, can kick an ISO up to 6400. Was that gonna be enough? Would I, for instance, be able to see an individual dino fire off?
That, for one, would take some very high magnification, but the microscope was probably out. I can’t say for sure even after my experiments, but it appears that the illumination might be exhausted for a short while after a burst, and the agitation of getting one onto a slide might well be enough – even using the pipette very gently, I could always see them glowing while traveling up the tube is if was dark enough. While within the orb, the amount of space for the dinos to float in would mean most of them being well out of focus. So what I needed was a very narrow space – still with good visibility.
 My solution was a new macro aquarium, made by sandwiching two pieces of clear acrylic together and then cutting them into a U shape, as seen here. This made a 4mm thick spacer, which would go between two more sheets of ‘full’ size, making a deep narrow trough to put water in. The back was the same acrylic, but for the front I used just a piece of glass from a 4×6″ photo frame from a discount store; acrylic is terrible about scratches and cloudiness, and that’s okay for the back surface but the front should be clearer. This is just a larger version of one that I made with slide glass many years ago. Provided that you have the materials and, like, a motor tool with a few different heads for that shaping, this doesn’t take too long. Everything was put together with cyanoacrylate (“super glue”) because the surfaces were all clean and flat. The seams proved to be perfectly waterproof, so then I was ready to wait until nightfall for the experiments.
My solution was a new macro aquarium, made by sandwiching two pieces of clear acrylic together and then cutting them into a U shape, as seen here. This made a 4mm thick spacer, which would go between two more sheets of ‘full’ size, making a deep narrow trough to put water in. The back was the same acrylic, but for the front I used just a piece of glass from a 4×6″ photo frame from a discount store; acrylic is terrible about scratches and cloudiness, and that’s okay for the back surface but the front should be clearer. This is just a larger version of one that I made with slide glass many years ago. Provided that you have the materials and, like, a motor tool with a few different heads for that shaping, this doesn’t take too long. Everything was put together with cyanoacrylate (“super glue”) because the surfaces were all clean and flat. The seams proved to be perfectly waterproof, so then I was ready to wait until nightfall for the experiments.
 Why night, you ask? Ah, you didn’t go to that link on the species name, did you? Dinoflagellates will only illuminate at night, governed by their circadian rhythms, so just getting into a dark place wouldn’t work (which may explain why my experiments with an aquarium filled with lagoon water in Florida failed.) I gently transferred some water into my mini aquarium with a pipette, then set up the camera rig.
Why night, you ask? Ah, you didn’t go to that link on the species name, did you? Dinoflagellates will only illuminate at night, governed by their circadian rhythms, so just getting into a dark place wouldn’t work (which may explain why my experiments with an aquarium filled with lagoon water in Florida failed.) I gently transferred some water into my mini aquarium with a pipette, then set up the camera rig.
I did several experiments to see what would work best. I wanted as much light as possible, so the reversed Sigma 28-105 (permanently set to f16) was out. I pulled out my old Sigma 105mm macro, because that was an f2.8 lens with a failed aperture (so, permanently at f2.8) and also the bellows with an Olympus 50mm f1.4 attached. I used a 36mm extension tube with the Sigma, and of course the bellows was significantly extended, so both of these cut down the light transmitted, but they were necessary to get the magnification needed to see an individual dino. The image below, by the way, is a full-frame shot through the sides of the orb with the Sigma macro and extension:

… while below is a nearly-full resolution inset of the same frame:
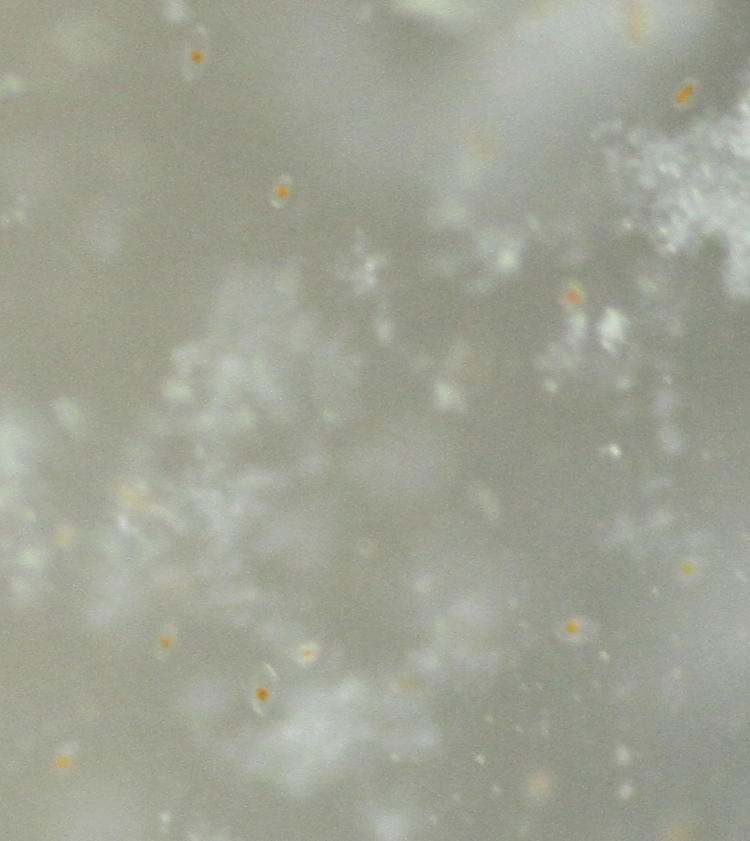
You can tell the dinoflagellates by their little orange centers. Not sharp of course, but close enough to see if any individual fired off at least.
So how did this all work?
Well, we’ve established that the brief burst of light can be captured on video, at least, though whether this will be possible at a high-enough magnification to see more detail than this is still in question. They need to be agitated to illuminate, so that generally means movement because my insulting their mothers hasn’t worked yet. So even if I have one pinned in the center of the frame in focus (which is what I was hoping for here,) the act of disturbing the water drives them out of the frame. There remains a chance that I could get one on a slide without exhausting its lighting ability (and I’m not even sure this is a limiting factor,) and then tapping the stage; I’m not holding my breath, but it might work. I may also try driving them down a clear plastic tube. But since I’ve been meaning to get this together for months now, I did at least make some progress on a project this winter, as well as preparing two macro aquariums (you have yet to see the other) for future uses. So there’s that.
* ADDENDUM: I linked to a source above, likely the one I received, but bioluminescent dinoflagellates are available from other sources not anywhere near as expensive. For instance, Carolina Biological Supply offers a living culture in comparable quantity for only $8.95 plus shipping (which is slightly pricey because these are biological specimens and have to ship 2nd Day or less.) Still, that’s a lot less than the above link, though you admittedly don’t get the classy orb and octopus stand. CBS is just down the road from me here, so I can just drive over for a new batch.