Okay, here’s the backstory. For some time now, I’ve been thinking of noodling around with photomicrography – photography through a microscope, you know, serious macro work. This requires having a microscope – surprise surprise – and I’ve been watching for one for years, since I wasn’t going to drop the serious money for a new one for something that I might not get into all that much. The local college surplus store had a few at one point several years ago, right at the edge of my price range, and I dithered and lost out, since they all went within two days. Since then, I never saw a decent microscope for sale anywhere.
Then not quite a year ago, I happened across one in a thrift store, complete except for the lighting unit, a serious four-objective binocular lab scope from Bausch & Lomb, which means price range new would be in the $500-$1000 range. Price on this unit? Eleven bucks. I actually went to check out with some trepidation, thinking they were gonna catch their mistake or even accuse me of switching price tags, but I walked out the door at that price. I can live with that.
Replacement light sources get expensive too, and I wasn’t finding a match anywhere, but we’re at this really cool point in tech now, and I simply fitted an LED light source. First unit burnt out within five minutes during the initial experiments, telling me that a heat sink is necessary immediately, but it was too bright anyway. Second unit was meticulously constructed to fit into the microscope housing with a generous heat sink, even feeding into the metal body of the scope – snazzy little job if I say so myself.
Finally, the other night I fired it up and started playing, finding that it was working just ducky. Then I couldn’t find the camera adapter that I’d purchased some time back. Located it the next morning, and later on tried again. I’m clearly going to need some slide cover slips and perhaps a top light source as well (the only one right now is the standard bottom light,) but you might start seeing more stuff like this (only better):
These came from a couple of different sessions as I experimented, just noodling around right at the moment, but proof of concept and all that rot. I also have to do a meticulous cleaning of the lenses all through. At some point, I’m going to figure out how to get a measuring scale in there – I imagine that they can be purchased someplace, and that they’re not cheap.
The other thing that appears in the middle clip is, I believe, a molted exoskeleton from another unidentified arthropod, found floating on the water sample – I included it as another subject to practice on, but did not explain this in the audio. All of these are from water drops merely resting on a slide, no cover slips or preparations or anything.
By the way, you can compare this with my results from using a bellows unit in place of a standard lens, several years back – daphnia come in a variety of sizes so I cannot say these are a direct comparison.
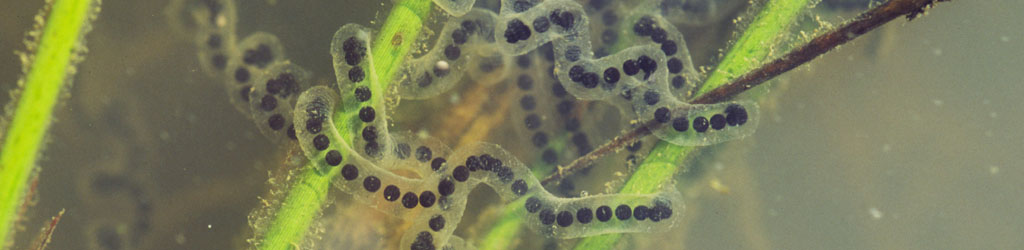
What prompted all of this was searching for something to photograph during the lean months, and doing a little poking around in the backyard pond. I turned up an egg case of some kind on the underside of a floating leaf, suspecting that they were snail eggs but honestly not sure. The remaining photos are all more-or-less ‘routine’ macro, taken with the reversed Sigma 28-105.

The entire blob was about 10mm in length, so you can figure out from there how small the eggs were. I include a detail inset from the same frame so you can see those eggs as well as we’re able right now (save for attempting to slice open the blob and get an individual egg on a slide, which i considered more likely to fail than succeed, but I also thought about getting some hatching sequences and so wanted to leave the blob undisturbed.)

There! That’s certainly… multicellular. If you can identify the species from those misshapen blobs, let me know – I think someone scrambled those eggs in the shell, myself.
After taking these, I returned the leaf to a small bucket where I could monitor things, but was unable to check back as routinely as I would have liked, and it appears they all hatched out in the interim. I still suspect snails, given how many are in the pond itself, but haven’t ruled out other things like that wormlike something that appeared first in the video.
One of the water samples that I gathered had a ride-along, a small insect that perched nonchalantly on the water surface and made no attempt to leave the 20ml sample jar that I was using, so I did a few shots of that one while it was being so cooperative.

I had no idea how to start looking for identification images, knowing this was not a truly aquatic bug, just one that was at home on the surface, so I uploaded the images to BugGuide.net, always a useful resource. But as I did so, I was looking at the anatomy again and realized it was like a short, squater version of a common water strider. In moments, I’d confirmed that this was indeed what it was, though not an immature nymph as I’d suspected from the lack of wings or elytra. The subsequent reply from BugGuide’s volunteers reinforced this discovery: it’s from the genus Microvelia. In this image it is cleaning its proboscis – striders are predatory hemipterans.
The very first things to appear, usually well before winter has gasped its last, are of course the daffodils, and there are only so many ways you can photograph them, but then again, what else is there? So when The Girlfriend had cut a few to put in a vase, I borrowed them one night to at least keep my hand in with the macro rig – I was beginning to think I’d forget what I was doing.

No no, of course that’s not where I stopped. Please.

Nothing like a straight-up-the-middle shot, eh? That stigma all moist and receptive, surrounded by eager ‘one-eyed’ anthers crowding around. I’ll let you pick your own appropriate soundtrack music…
I remembered something from my childhood, one of those elementary school experiments that are supposed to show – something, who knows what – but mostly just look cool. One of those things that you read or hear about, but never actually try, you know? So, given that I’m in my mid-fifties now, I figured it was time to get to this long-neglected task, and cut a new flower to put in its own vase on my desk, adding a few drops of food coloring to its water.
The idea is that the flower draws up the water for a few days and the food coloring stains its petals, creating unnatural hues in the blossoms (it supposedly also works with celery.) My plan was to get it fairly unique in color and then sneak it into The Girlfriend’s own vase and see how long it took her to notice. But perhaps daffodils are the wrong choice for this, because while some coloration did indeed occur, it was to a very limited extent before the flower dried up, too subtle to be of any real use. Ah well.

Given those, you should now know what the end of the month abstract image was; as the flower started to dry out, I took a section of a petal to pop under the microscope, naturally picking a portion that was showing the color. And it might be showing some evidence of this, if you look closely at the image at that link – there are faint hints of cyan here and there, but that could also just be refraction of the light too. Still, I’m just getting started, so we’ll see what kind of funky stuff I can dig up later on.